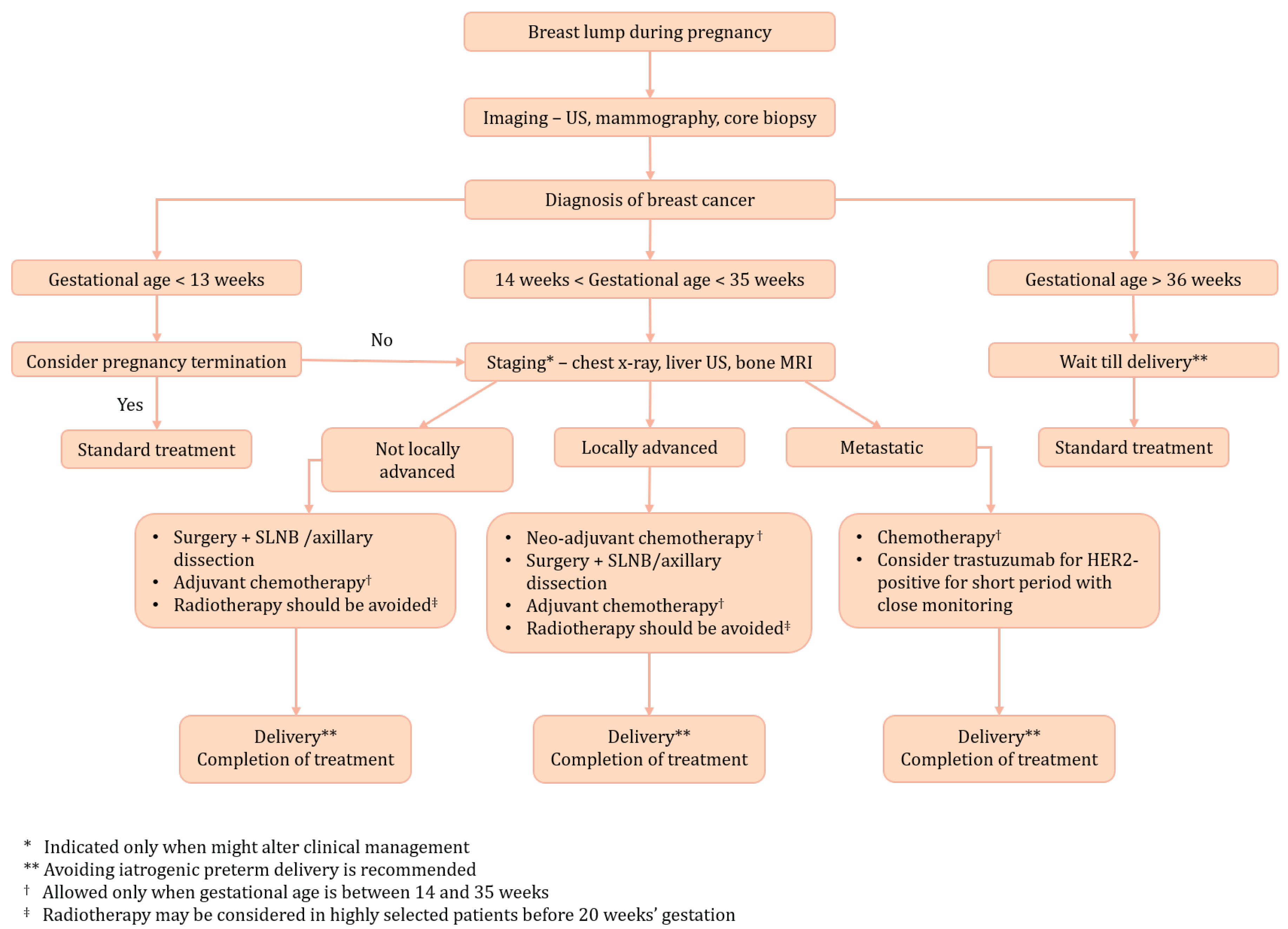
An investigation into the performance of the Adjuvant! Online prognostic programme in early breast cancer for a cohort of patients in the United Kingdom | British Journal of Cancer
Risk Estimations and Treatment Decisions in Early Stage Breast Cancer: Agreement Among Oncologists and the Impact of the 70-Gene Signature - Agendia Inc.

Adjuvant S-1 plus endocrine therapy for oestrogen receptor-positive, HER2-negative, primary breast cancer: a multicentre, open-label, randomised, controlled, phase 3 trial - The Lancet Oncology

An investigation into the performance of the Adjuvant! Online prognostic programme in early breast cancer for a cohort of patients in the United Kingdom | British Journal of Cancer

Pathologic evaluation of response to neoadjuvant therapy drives treatment changes and improves long‐term outcomes for breast cancer patients - Bossuyt - 2020 - The Breast Journal - Wiley Online Library

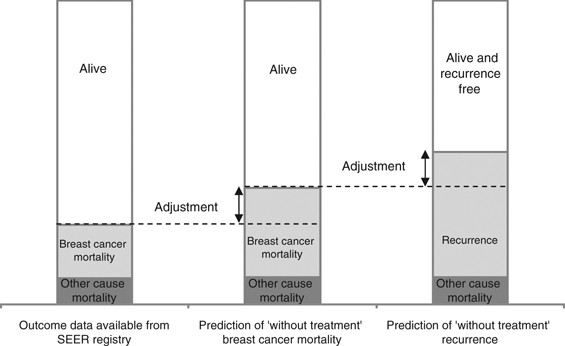
PDF) External Validation of Adjuvant! Online Breast Cancer Prognosis Tool. Prioritising Recommendations for Improvement | Anne Vincent-salomon - Academia.edu

Extended adjuvant aromatase inhibition after sequential endocrine therapy in postmenopausal women with breast cancer: follow-up analysis of the randomised phase 3 DATA trial - eClinicalMedicine

Risk for breast cancer recurrence as determined by Adjuvant! Online and... | Download Scientific Diagram

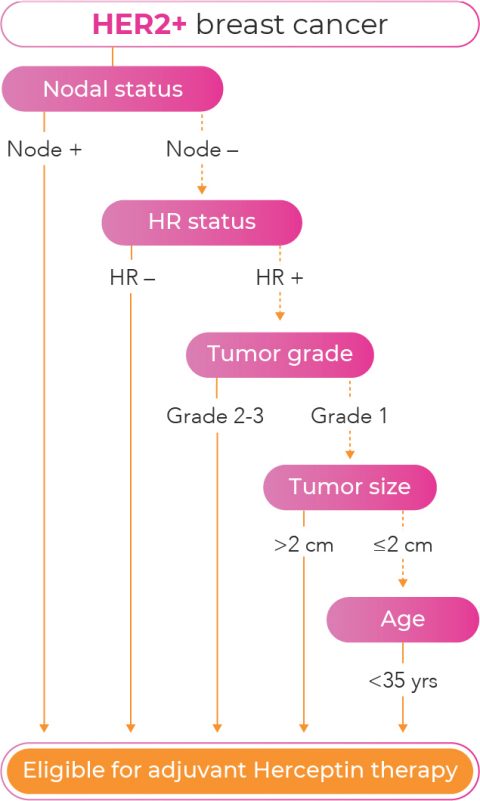
Role of Patient and Disease Factors in Adjuvant Systemic Therapy Decision Making for Early-Stage, Operable Breast Cancer: Update of the ASCO Endorsement of the Cancer Care Ontario Guideline - Agendia Inc.

Prolonged Time to Adjuvant Chemotherapy Initiation Was Associated with Worse Disease Outcome in Triple Negative Breast Cancer Patients | Scientific Reports

Anthracycline-free or short-term regimen as adjuvant chemotherapy for operable breast cancer: A phase III randomized non-inferiority trial - The Lancet Regional Health – Western Pacific
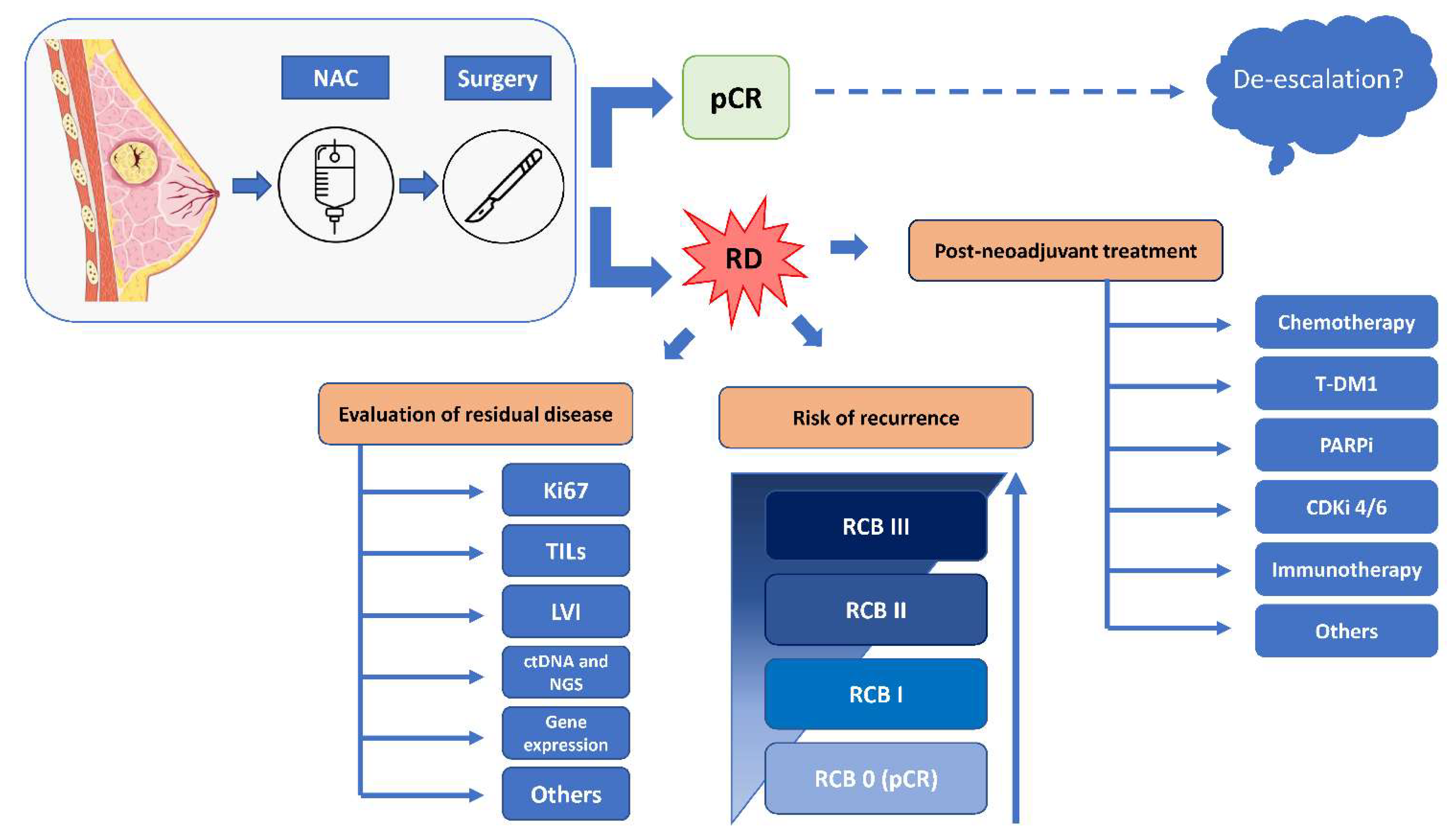
Cancers | Free Full-Text | Post-Neoadjuvant Treatment Strategies for Patients with Early Breast Cancer